Smaller proteins migrate faster than larger ones due to less resistance from the gel until pockets within the gel encapsulating proteins of similar size and charge are produced. In this method, an electric field forces proteins through the pores of a gel. Like many characterization techniques, western blotting uses gel electrophoresis to separate proteins based on size and charge. The CBB-stained gels were photographed using transmitted white light without an optical filter. The ProLite™ Orange stained gels were photographed using a SYPRO Orange filter. Three-fold dilution series of BSA standards were separated on a NuPAGE ® 4-12% Bis-Tris gel and stained with A) ProLite™ Orange Protein Gel Stain or B) Coomassie brilliant blue (CBB) according to standard protocols. With the development of novel blotting technologies and specialized automated equipment, western blotting has become a cornerstone in proteomics research and protein detection, producing qualitative and semi-quantitative results. Western blotting is used extensively in biochemistry applications targeting complex proteins, testing for disease markers (e.g., HIV, Lyme disease, or Hepatitis B), and confirming protein production in cloning experiments. Common reporter molecules include enzymes, such as horseradish peroxidase (HRP) and alkaline phosphatase (ALP), or fluorescent dyes, such as iFluor® 488, with the latter affording greater sensitivity and multiplexing capacity. Antibodies are selected based on their specificity for the protein of interest, and this specificity of the antibody-protein interaction enables target identification. This process involves the electrophoretic separation of proteins, the transfer of separated proteins from a gel to a stable membrane substrate, and their subsequent detection by antibodies labeled with reporter molecules. The ladder's probably fine, but if these are cell lines or mouse tissue you should bump it up a bit (assuming your 1Abs are high affinity and specificity for your target and are generally doing their job well).Western blotting is an analytical technique used to detect the presence of specific proteins in a complex biological sample. Also, are you loading enough protein per well? The signal looks extremely faint and I can really only make out 2 sample lanes. Then, I'd work backwards and make sure it's not my ECL reagent (is it fresh?), my 2Abs (is the concentration high enough to get a good signal?), my 1Abs (are these high affinity enough for my target? What happens if I double or half my usual concentration?), my transfer (did I transfer for long enough and did all my protein get transfered? I'd do a Ponceau or check that all dye left the gel in the case of visual dyes in the molecular ladder), and finally my gel running settings. Because it's easy, I would maybe first play around with the length of exposure on the chemidoc and just make sure the issue isn't there first. Voltage during the run, if too high, can definitely lead to smearing in the high molecular weight range, band "smiling" or other artifacts.  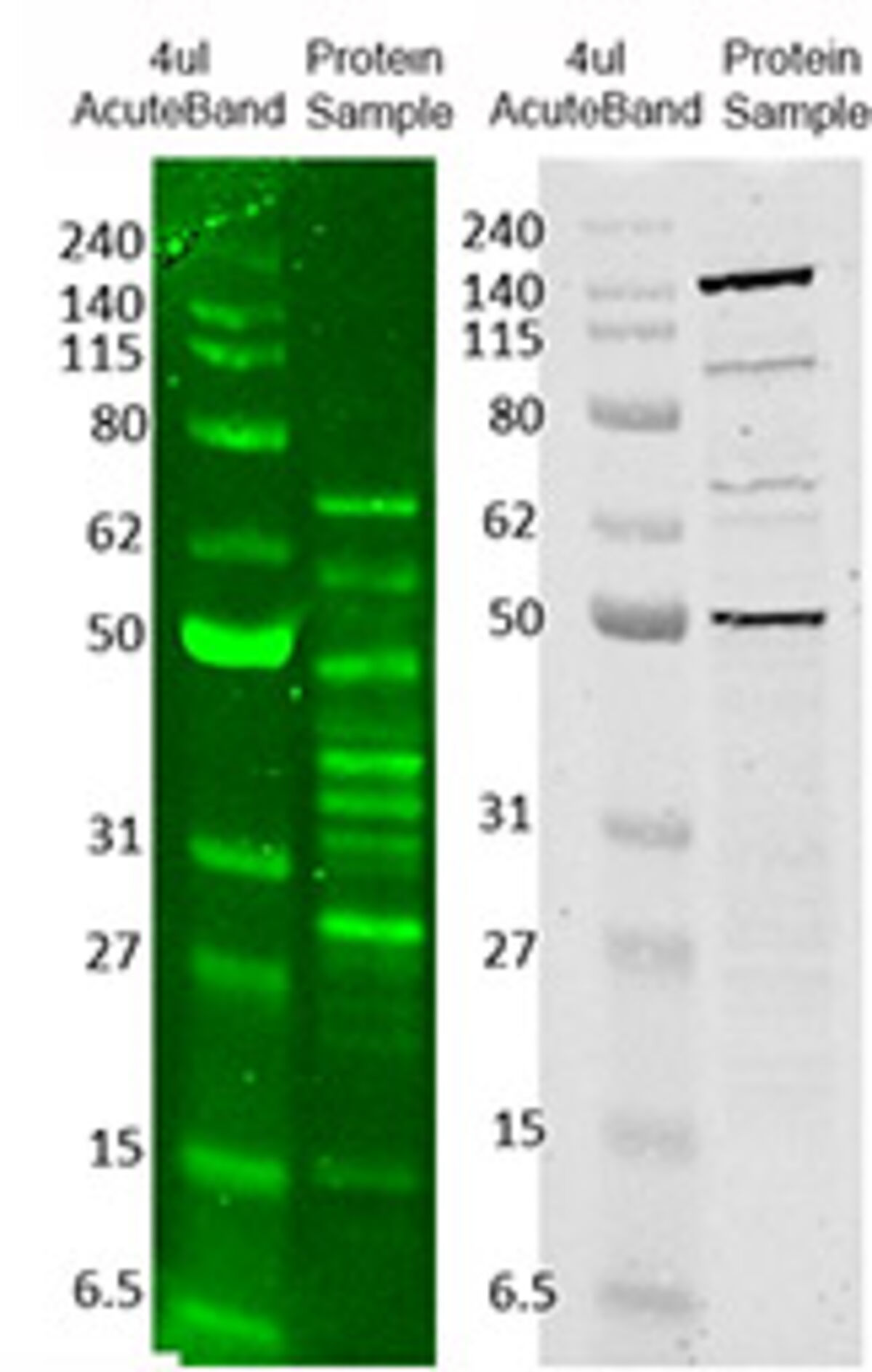
You may want to see if lowering the voltage a bit or switching running buffers makes a difference for the stacking and tightness of the ladder bands. Okay that helps a bit! Regarding the ladder, I assume it's detected via HRP? Or is it a proprietary BIO-RAD technology that allows it to be detected in some other wavelength range? IMO it's a little blurry and it could be stronger.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
